In 1913, electricity still felt like magic to much of the world.
Homes were only beginning to glow with electric light. Radios were rare curiosities. Telegraph wires stitched towns together like veins carrying invisible signals. And for countless boys and hobbyists across America, the dream of harnessing this mysterious force often began on a kitchen table.
Old newspapers and magazines from the early twentieth century were filled with strange instructions for homemade science projects — crystal radios, telegraph sets, lightning experiments, and even dangerous chemical batteries assembled from jars, carbon rods, acid, and scrap metal.
To modern eyes, many of these experiments feel reckless. To readers in 1913, they represented progress.
One newspaper article published that year taught young readers how to build their own electric batteries at home using sal-ammoniac, sulphuric acid, mercury, and discarded battery parts.
The article reads like something between a chemistry lesson and a survival guide from a forgotten age of experimentation.
Below is the original article as it appeared in the Vilas County News on August 20, 1913.

Homemade Electric Batteries

Every boy who experiments in electrical work should know how to make the common forms of battery cells. They are simple to make, and the materials are inexpensive and easily obtained.
There are two general classes of batteries — those made for “open circuit” work, such as electric bell circuits and telegraph circuits, where the current is not drawn upon very long at a time, and those made for “open circuit” work, such as for operating small motors, induction coils, etc., where there is more or less of a steady drain upon the current.
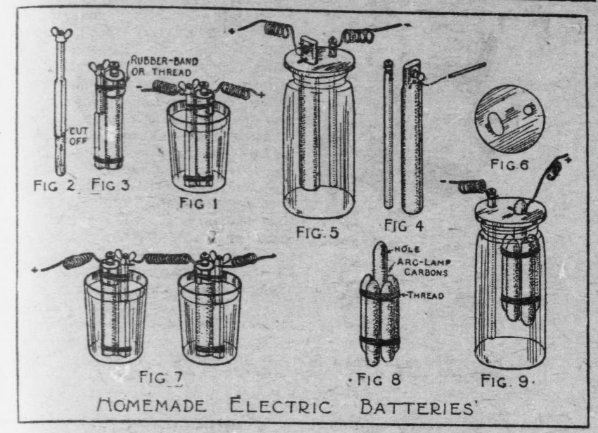
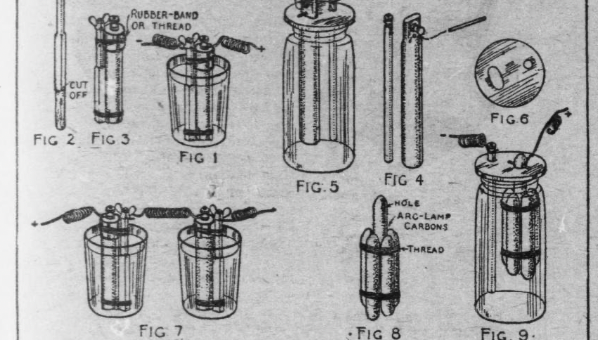
The sal-ammoniac battery is the simplest of the open circuit type. A glass tumbler will do for a jar, an old battery zinc pencil with several inches of the eaten end cut off will do for the zinc element (Fig. 2), and the carbon from a worn out dry battery, cut to a corresponding length, will do for the carbon element.
Fasten together the zinc and carbon with rubber bands, as shown in Fig. 3, after wrapping a piece of bicycle tape around the upper end of the carbon, and inserting a small wad of it between the lower ends of the carbon and zinc, to keep them touching each other.
Make a solution of 8 ounces of sal-ammoniac to a quart of water, or 4 ounces to a pint.
This battery will not be strong enough for more than simple experimental work.
For a stronger battery, make two cells and connect the two in “series;” that is, connect the zinc pole of one with the carbon pole of the other, as shown in Fig. 7.
A quart size Mason fruit jar, or a large tobacco jar or wide necked pickle bottle, can be used for a larger battery cell. Use a full length zinc pencil and old dry battery carbon for the elements (Fig. 4 and 5). Cut a round or square block of wood for a top to the jar (Fig. 6) and make holes in it for the ends of the zinc and carbon to stick through.
If you use a zinc similar to the one shown in Fig. 4, the little lugs upon its sides will be sufficient to support it.
To support the carbon, drilling a hole through it just below the connection thumb nut, with the point of a nail, and slip a match through the hole (Fig. 5).
Another way of providing for the carbon element is shown in Fig. 7. Pick up some arc lamp carbon ends from around the street lamp poles and remove the copper coating from them with a file or sandpaper. Then bind five of the pieces around a center piece, as shown in Fig. 8, allowing the upper end of the center piece to project about 1-1/2 inches above the others. With the point of a nail, drill a hole through the center carbon just below the top, through which to stick a match for support (Fig. 9). The connection wire is also run through this hole.
The bi-chromate batter is the simplest type of closed circuit batteries to make. The carbon and zinc elements of this may be the same as those used for a sal-ammoniac cell. And a tumbler or Mason fruit jar may be used to hold the battery fluid.
The bi-chromate solution is made up of bi-chromate of potash, sulphuric acid and water in the following proportions:
Four ounces bi-chromate of potash.
Four ounces sulphuric acid.
One quart of water.
In making up the solution, first add the acid to the water — not the water to the acid — and then when the solution has almost cooled, add the bi-chromate of potash.
Pour the acid slowly into the water, because the combination creates a great deal of heat, and if the heat forms too quickly your glass bottle is likely to split.
Label the bottle in which you put this solution POISON.
The bi-chromate solution attacks the zinc element even when the current is not being drawn upon, so the zinc should be removed when the battery is not in use.
To reduce the eating of the zinc to a minimum, it should be amalgamated by rubbing a thin coat of mercury over its surface. Dip the zinc into the solution first, then with a rag dipped in the solution, rub the mercury onto it.
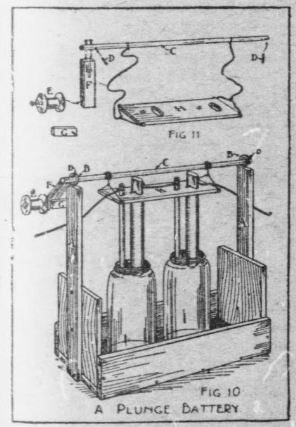
The plunge battery shown in Fig. 10 contains two bi-chromate cells, with an arrangement by which the elements of the cells can be raised out of, and lowered into, the solution.
A grocery box can be cut down to a convenient size for the rack. Nail the uprights A (Fig. 10) to the ends of the rack to support the winding drum C, and make them of the right length so C will be the length of the carbon above the battery jars.
Screw screw-eyes into the tops of the uprights A for drum C to turn in (B, Fig. 10), and bore a hole through C near each end to receive the wooden pins D (Fig.11), which prevent the drum from slipping through the screw-eyes.
A spool forms the crank handle E, and this is pivoted with a nail to the crank F, and the end of the drum is nailed to the end of the crank (Fig. 11).
The button G is screwed to one edge of the upright A, at the crank end, and when turned to the position shown in Fig. 10 it checks the crank.
The board H supports the battery elements, and the raising cord is attached to nails driven into its edges and the drum C.
Source: Vilas County News. Eagle River, Vilas County, Wis. August 20, 1913.
You can find Neely Hall’s book, Handicrafts for Handy Boys, on Amazon. (Linked)


